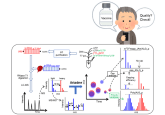
Case study: automating cell-based assays to determine viral vector titer
In this case study, Oxford Biomedica (Oxford, UK), an innovative global gene and cell therapy company with operations in the UK and US, describes the automation of a 10 day cell-based workflow required for an analytical test to determine viral vector titer, using Hamilton Microlab® STARTM liquid handlers.
Highlights of this case study include:
- Detailed description of the automated system used for the cell-based workflow
- Advantages of Hamilton’s CECULA Graphical User Interface (GUI) for the planning of long-term cell-based assays
- Transfer of the automated assay to Good Manufacturing Practice (GMP)
In association with